Basic concepts: molecular biology, DNA, RNAs, chemical bonds, importance of water, chemical reactions, organic compounds, carbohydrates, lipids, cholesterol, proteins, amino acids, fats, steroids, emulsifiers, etc.
Disclaimer: While every reasonable effort is made to ensure that the information provided is accurate, no guarantees for the currency or accuracy of information are made. It takes several proof readings and rewrites to bring the quiz to an exceptional level. If you find an error, please contact me as soon as possible. Please indicate the question ID-Number or description because server may randomize the questions and answers.
Go to: Midterm I | Midterm II | Final Exam
Biology 205 (BIOL 205-UCAL) Midterm III
Congratulations - you have completed Biology 205 (BIOL 205-UCAL) Midterm III.
You scored %%SCORE%% out of %%TOTAL%%. With incorrect multiple attempts your score is %%PERCENTAGE%%
Your performance has been rated as %%RATING%%
Question 1 |
A | α−1-->4 |
B | β−1-->1 |
C | β−1-->4 |
D | α−1-->1 |
Question 2 |
A | RNA has Uracil (Ura) nitrogenous base instead of Thymine (Thy). |
B | RNA has Cytosine (Cyt) nitrogenous base instead of Adenine (Ade). |
C | RNA has Thymine (Thy) nitrogenous base instead of Cytosine (Cyt). |
D | RNA has Thymine (Thy) nitrogenous base instead of Adenine (Ade). |
E | RNA has Adenine (Ade) nitrogenous base instead of Cytosine (Cyt). |
F | RNA has Guanine (Gua) nitrogenous base instead of Cytosine (Cyt). |
Question 3 |
Note: Moose dung is a traditional Canadian delicacy. Yum yum 🙂
A | Increase the humidity of the cook pot. |
B | Decrease the humidity of the cook pot. |
C | Freeze the dung before cooking. |
D | Add salts, ionic compounds, to the moose dung. |
Question 4 |
A | Nucleic Acids |
B | Proteins |
C | Carbohydrates |
D | Lipids |
Question 5 |
A | 65% |
B | 75% |
C | 100% |
D | 50% |
E | 99% |
Question 6 |
I. Covalent bonds are stronger than ionic bonds.
II. Chemical reactions always result in loss of electron(s).
III. Water is slightly polar.
IV. Ionic bonds are bound together with oppositely charged ions.
A | I and III |
B | I , II and III |
C | I and II |
D | II and IV |
E | I , III and IV |
F | All of the above statements are correct. |
Question 7 |
A | 8 electrons. |
B | 5 electrons. |
C | 13 electrons. |
D | 5 electrons. |
E | 10 electrons. |
Question 8 |
A | Process in which chemical difference between inside and outside the cell is maintained. |
B | Production of proteins by the bone marrow to keep stable white blood cell count. |
C | A digestive process used by herbivorous to break down the plant cells. |
D | Movement of an organism in response to a chemical stimulus. |
Question 9 |
A | Organelles |
B | Nucleus |
C | Cell walls |
D | DNA as genetic materials |
Question 10 |
A | 4 |
B | 2 |
C | 6 |
D | 5 |
E | 3 |
Question 11 |
A | Nitrogen and Oxygen |
B | Carbon and Hydrogen |
C | Hydrogen and Oxygen |
D | Hydrogen, Oxygen and Nitrogen |
E | Carbon, Oxygen and Nitrogen |
F | Hydrogen, Carbon and Oxygen |
Question 12 |
A | 1.0 to 10 micrometres |
B | 0.1 to 1.0 micrometres |
C | 100 to 1000 micrometres |
D | 10 to 100 micrometres |
Question 13 |
A | 20 |
B | 100 |
C | 35 |
D | 50 |
E | Millions |
Question 14 |
A | Temperature is the amount of energy associated with movement of atoms while heat is the intensity of heat (average speed of atoms). |
B | Temperature is a long term quantitative measurement of energy while heat is a short term measurement of energy. |
C | Heat is the amount of energy associated with movement of atoms while temperature is the intensity of heat (average speed of atoms). |
D | Heat is produced through instantaneous reactions such as boiling water. Temperature is the quantitative measurement of heat. |
Question 15 |
A | There is a world wide shortage of advanced microscopes. |
B | Movement of organic material is magnified in advanced microscopes hence harder to study. |
C | Light microscopes are cheaper for a biologist to obtain. |
D | The techniques used to study microscopic particles in cell biology has not changed since 1665. |
Question 16 |
A | 1/50th of a typical prokaryotic celll. |
B | 1/30th of a typical prokaryotic celll. |
C | 1/100th of a typical prokaryotic celll. |
D | 1/10th of a typical prokaryotic celll. |
E | 1/25th of a typical prokaryotic celll. |
Question 17 |
A | 4 |
B | 2 |
C | 3 |
D | 5 |
Question 18 |
A | Scanning Electron Microscope (SEM) |
B | Electron Microscope (EM) |
C | Light Microscope (LM) |
D | Transmission Electron Microscope (TEM) |
Question 19 |
A | Carbonyl group |
B | Carboxyl group |
C | Phosphate group |
D | Amino group |
E | Hydroxyl group |
Question 20 |
A | ATP injection |
B | Hydrolysis |
C | Heating |
D | Protein pumps |
Question 21 |
A | ...unsaturated fats. |
B | ...good for human health/digestive system. |
C | ...solid at room temperature. |
D | ...contain more nutrition than plant based fats. |
Question 22 |
A | Very high reaction rate. |
B | The chemical properties such as high cohesion and polarity. |
C | The physical properties such as very low angles between the two H-atoms and the O-atom. |
D | Very high electron transfer rate and high electrical conduction rate. |
Question 23 |
A | ...the type of translation. |
B | ...water solubility level. |
C | ...their orientation of the hydrogen atoms. |
D | ...their chemical structures. |
Question 24 |
A | Low radioactivity and polarity. |
B | The high atomic mass. |
C | The hydrogen bonds. |
D | The very high electronegative nature of the molecule. |
Question 25 |
A | I. covalent bonds II. nucleic bonds |
B | I. ionic bonds II. peptide bonds |
C | I. covalent bonds II. peptide bonds |
D | I. ionic bonds II. nucleic bonds |
Question 26 |
A | Fructose is cheaper than glucose. |
B | Fructose is a polymer while glucose is a monomer. |
C | There is more fructose naturally in corn syrup. |
D | Fructose is sweeter than glucose. |
Question 27 |
A | All organic matter is compounds while molecules are inorganic. |
B | Molecules composed of two or more different elements while compounds composed of two or more atoms. |
C | Compounds composed of two or more different elements while molecules composed of two or more atoms. |
D | Molecules are always ionic while compounds could be either ionic or covalent. |
E | Molecules easily react with organic matter therefore they often involved in chemical reactions. Compounds are stable entities and are often inert. |
Question 28 |
A | They are only found in plant cells because animal cells with movement cannot maintain the structure. |
B | They are composed of action and involved in cell movement and maintaining cell structure. |
C | They are composed of globular proteins that are assembled and disassembled in different regions of the cell to form hollow pathways. |
D | They made the various fiberous proteins of α-helical
coiled-coils that transport materials from one area to another. |
Question 29 |
A | Nucleolus |
B | Vesicles |
C | Rough endoplasmic reticulum |
D | Golgi apparatus |
E | Centriole |
Question 30 |
A | ...higher the radioactivity of the atom. |
B | ...more stable the atom. |
C | ...higher the pull of electrons towards its nucleus. |
D | ...lower the radioactivity of the atom. |
E | ...lower the pull of electrons towards its nucleus. |
Question 31 |
A | Red blood cells |
B | White blood cells |
C | Platelets |
D | Serum |
Question 32 |
A | They are nonploar and therefore hydrophilic. |
B | They can form many different shapes with random orientations. |
C | They do not form polymers. |
D | They are the largest biological molecules. |
Question 33 |
A | To be able to process food and data. |
B | To allow nutrients and gasses to pass across the cell surface. |
C | To be able to stack together to form tissues. |
D | To be able to communicate to the outside world. |
Question 34 |
A | ...Plasma membrane, Ribosomes and Lysosome. |
B | ...Lysosome and Centriole. |
C | ...Microtubule, Intermediate filament and Microfilament. |
D | ...Ribosomes, Lysosome and Centriole. |
E | ...Centriole, Golgi apparatus and Ribosomes. |
Question 35 |
A | They often form ring structures. |
B | Most common two; glucose forms 5 membered structures while fructose forms 6 membered structure. |
C | Their molecular formulas are usually multiples of CH2O2. |
D | They usually characterized by three double bonded C-O structures. |
Question 36 |
A | disaccharide |
B | monsaccharide |
C | lipid |
D | polysaccharide |
E | protein |
Question 37 |
A | Some proteins only have α helix structures while others can have only β pleated sheet structures. |
B | Hydrogen bonds are primary structures of proteins. |
C | The α helix and the β pleated sheet structures are tertiary structure. |
D | Protein structures have four levels of structures. |
Question 38 |
A | The difference in pH levels inside and outside the cell facilitates movement of ionic and polar molecules. |
B | Receptor proteins codes the ionic and polar molecules so that it will be accepted by the phospholipids. |
C | Transport proteins form channels which allow ions and polar molecules to move across. |
D | Simple diffusion process. |
Question 39 |
A | Thorium gas |
B | Sulfur mustard gas |
C | Natural gas |
D | Radon gas |
Question 40 |
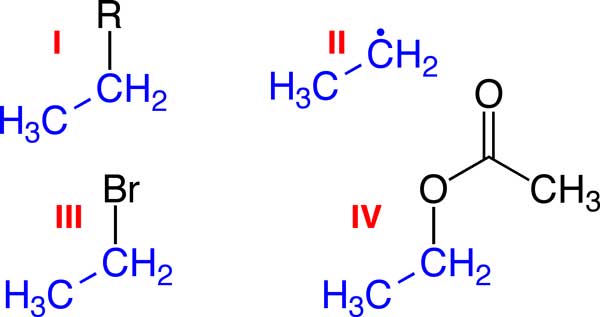
Image mod from: http://en.wikipedia.org/wiki/Ethyl_group
A | II and IV |
B | III only |
C | I, II and IV |
D | II only |
E | IV only |
F | I only |
Question 41 |
A | Carbonyl group |
B | Amino group |
C | Methyl group |
D | Hydroxyl group |
E | Carboxyl group |
F | Phosphate group |
Question 42 |
A | Primary structure |
B | Secondary structure |
C | Tertiary structure |
D | Quaternary structure |
Question 43 |
A | 22 , iron |
B | 25 , nitrogen |
C | 15, iron |
D | 15 , sodium |
E | 22 , sodium |
F | 15 , nitrogen |
Question 44 |
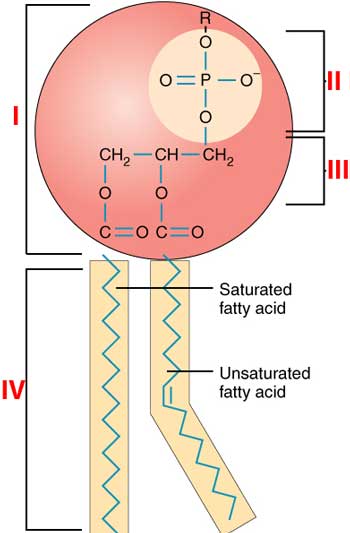
Image mod from: http://commons.wikimedia.org/wiki/File:0301_Phospholipid_Structure.jpg
A | III and I |
B | III and IV |
C | I and III |
D | IV and I |
E | I and IV |
Question 45 |
A | Theory of inheritance |
B | Genetic theory |
C | Cell theory |
D | Endosymbiont theory |
Question 46 |
A | More triglycerides within the lipoprotein, hence away from the blood. |
B | More cholesterol within the lipoprotein, hence away from the blood. |
C | A very low LDL to HDL ratio. |
D | A very high LDL to HDL ratio. |
Question 47 |
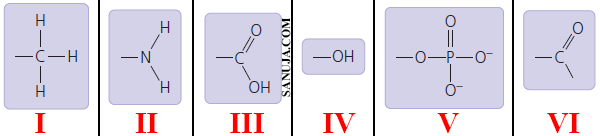
Suggestion: Take your time to answer this question. It can be confusing with 6 types!
A | VI , IV , III , II , V and I |
B | IV , VI , II , III , V and I |
C | I , III , IV , V , II and VI |
D | IV , I , VI , V , II and III |
E | IV , VI , III , II , V and I |
F | VI , IV , II , V , III and I |
Question 48 |
A | Number of electrons within the structure increases. Hint: Not always, but sometimes. |
B | Neutrons and protons from different atoms react with each other. |
C | Produce molecules and compounds with polarity. Hint: Not always, but sometimes. |
D | Valance shell electrons react with other atoms to form bonds. |
Question 49 |
A | 3 |
B | 2 |
C | 6 |
D | 5 |
E | 4 |
Question 50 |
A | The mixing of the two most likely have broken down the molecular bounds of water and that resulted in reaction with cocoa powder. |
B | The milk acted as the solute which breaks down the chemical compounds in the cocoa powder. |
C | The temperature of the mixture should have increased as the mixing progresses. |
D | Milk is the solvent while cocoa powder is the solute. |
Question 51 |
A | Butane |
B | Methane |
C | Ethane |
D | Benzene |
Question 52 |
A | 2 times more H+ |
B | 10 times more H+ |
C | 100 times more H+ |
D | 100 times less H+ |
E | 10 times less H+ |
[H+] = 10-pH
Question 53 |
A | Biological cells have the ability to differentiate Carbon-12 from Carbon-14. |
B | Majority of organic cells lacks nucleus. |
C | Only the animal cells that have materials that move around (mobile). |
D | Both animal and plant cells contains materials that move around (mobile). |
E | Natural carbon is only produced by plant cells. |
Question 54 |
A | 3-carbin sugar, phosphate group and a nitrogenous base |
B | 5-carbin sugar, phosphate group and a nitrogenous base |
C | 3-carbin sugar, phosphate group and a adenine |
D | 5-carbin sugar, phosphate group and a adenine Hint: While it is true a adenine can be one, it is not a general part! |
Question 55 |
A | DNA and RNA replication. |
B | Locomotion. |
C | Enzyme and proteins synthesis. |
D | Provide structural support. |
E | Protection from the outside environment. |
Question 56 |
A | Quaternary structure |
B | Tertiary structure |
C | Primary structure: |
D | Secondary structure |
Question 57 |
6 C? (ID-B03-04)
A | 7 |
B | 14 |
C | 8 |
D | 6 |
Question 58 |
A | The tRNA carries amino acids with high energy bound for making proteins. |
B | Coding of the proteins is done by mRNAs. |
C | Only the mRNA structure is linear. |
D | Only the tRNA structure is linear. |
Question 59 |
A | Provide energy for the DNA structure by converting energy from cells into ATP and transferring it to the nucleus. |
B | Protect the nucleus from genetic mutations, even though this always does not work. |
C | Develop new genetic codes for evolutionary adaptations. |
D | Carry information of amino acid sequence from the genes to make proteins in cells. |
Question 60 |
A | I. translation II. transcription |
B | I. replication II. translation |
C | I. replication II. transcription |
D | I. translation II. replication |
E | I. transcription II. translation |
F | I. transcription II. replication |
Question 61 |
A | Neutrons |
B | Protons and Electrons |
C | Protons |
D | Electrons |
E | Protons and Neutrons |
Question 62 |
A | ...always outside cell walls. |
B | ...in aqueous environment of cells. |
C | ...by heating the reactants. |
D | ...only by breaking covalent bonds because living cells cannot break down ionic bonds. |
Question 63 |
A | Fructose |
B | Maltose |
C | Glucose |
D | Cellulose |
Question 64 |
A | Starch |
B | Lipids |
C | Chitin |
D | Cellulose |
E | Glycogen |
Question 65 |
A | Forms cell membranes. |
B | Signaling molecules for sex hormones. |
C | Energy storage and release. |
D | Synthesis of Amino acids, DNA and RNA. |
Question 66 |
A | Genetic control of the cell. |
B | Energy processing. |
C | Provide structural support for the cell wall. |
D | Manufacture and distribution of molecules. |
E | Provide structural support for the nucleus. |
Question 67 |
A | Aquaporins protein |
B | Glycophorin protein |
C | Ankrin protein |
D | Band 3 protein |
E | Spectrin protein |
Question 68 |
A | ...obesity. |
B | ...high cholesterol. |
C | ...thyroid problems. |
D | ...diabetes. |
Question 69 |
A | Isolation from the outside; like a cell wall. |
B | Attaching to host surfaces. |
C | Facilitate active transport and diffusion. |
D | Locomotion. |
Question 70 |
A | Gap junctions |
B | Anchoring junctions |
C | Messenger junctions |
D | Tight junctions |
E | Membrane junctions |
Question 71 |
A | The base A pairs with base G while base C pairs with base T to form a double helix RNA. |
B | The base A pairs with base T while base C pairs with base G to form a double helix DNA. |
C | The base A pairs with base C while base G pairs with base T to form a double helix RNA. |
D | The base A pairs with base T while base G pairs with base T to form a double helix RNA. |
E | The base A pairs with base G while base C pairs with base T to form a double helix DNA. |
F | The base A pairs with base C while base G pairs with base T to form a double helix DNA. |
Question 72 |
A | Cellulose |
B | Starch |
C | Carboxylic acids |
D | Glycogen |
Question 73 |
A | Two subatomic particles. |
B | Hundreds of subatomic particles. |
C | Five subatomic particles. |
D | Three subatomic particles. |
Question 74 |
A | They process materials and produce usable energy for eukaryotic cells. |
B | They are the transport vesicles that deliver glycoprotein from ER membrane to Golgi apparatus. |
C | In humans, they acts as the digestive compartments within cells. |
D | In plants, they store toxins to protect them from predators. |
Question 75 |
A | I. chromosomes II. ribosomes |
B | I. ribosomes II. nucleoli |
C | I. ribosomes II. lysosomes |
D | I. chromosomes II. chromatin |
E | I. lysosomes II. nucleoli |
Question 76 |
A | Ribosomes, organelles and hemoglobin. |
B | Nucleus, plasma membrane and hemoglobin. |
C | Ribosomes, plasma membrane and hemoglobin. |
D | Plasma membrane and hemoglobin. |
E | Nucleus, ribosomes and hemoglobin. |
Question 77 |
A | Decrease production in glycoprotein. |
B | Increase in rough endoplasmic reticulum. |
C | Increase in smooth endoplasmic reticulum. |
D | Increase production in glycoprotein. |
Question 78 |
A | Glucose |
B | Chitin |
C | Flagella |
D | Peptidoglycan |
Question 79 |
A | They have kinks caused by double bonding of carbon atoms. |
B | They are often found in olive oil and vegetable oils. |
C | They have the maximum number of hydrogens (alkanes) in the fats and lipid group. |
D | Compared to unsaturated fatty acids, they have a very high melting point. |
Question 80 |
A | They slow down chemical reaction by acting as a buffer. |
B | They speed up chemical reactions without participating in the reaction. |
C | They speed up chemical reactions by participating in the reaction. |
D | They slow down chemical reaction by participating in the reaction. |
Question 81 |
A | Chemical precipitation reactions |
B | Hydrogen bonding |
C | Vital hormone producing reactions |
D | Acid-base reactions |
Question 82 |
A | The elemental abundance must be less than 0.1% of the human body weight. |
B | The elemental abundance must be less than 0.2% of the human body weight. |
C | The elemental abundance must be less than 1.0% of the human body weight. |
D | The elemental abundance must be less than 0.4% of the human body weight. |
E | The elemental abundance must be less than 0.01% of the human body weight. |
Question 83 |
A | Laws of inheritance |
B | Genetic theory |
C | Molecular theory |
D | Theory of evolution |
E | Cell theory |
Question 84 |
A | False |
B | True |
Question 85 |
A | False |
B | True |
Question 86 |
A | Tight junctions |
B | Gap junctions |
C | Fiber junctions |
D | Anchoring junctions |
Question 87 |
A | Because CO2 reacts with calcium to produce bicarbonate. |
B | Because CO2 reacts with water to produce carbonic acid. |
C | Because CO2 is an acid. |
D | Because CO2 acts as a low pH buffer. |
Question 88 |
A | Salivary gland |
B | Thyroid gland |
C | Prostate gland |
D | Pituitary gland |
E | Pineal gland |
Question 89 |
A | A substance that prevent genetic mutations. |
B | A substance that increase the pH level of a high pH solution while decrease the pH level of a low pH solution. |
C | A substance that resists genetic mutations. |
D | A substance that prevent changes in pH by accepting or donating hydrogen irons. |
E | A substance that resists changes in pH by accepting or donating hydrogen irons. |
Question 90 |
A | Carbon-12 |
B | Helium-4 |
C | Carbon-14 |
D | Nitrogen-12 |
E | Carbon-13 |
← |
List |
→ |
| 1 | 2 | 3 | 4 | 5 |
| 6 | 7 | 8 | 9 | 10 |
| 11 | 12 | 13 | 14 | 15 |
| 16 | 17 | 18 | 19 | 20 |
| 21 | 22 | 23 | 24 | 25 |
| 26 | 27 | 28 | 29 | 30 |
| 31 | 32 | 33 | 34 | 35 |
| 36 | 37 | 38 | 39 | 40 |
| 41 | 42 | 43 | 44 | 45 |
| 46 | 47 | 48 | 49 | 50 |
| 51 | 52 | 53 | 54 | 55 |
| 56 | 57 | 58 | 59 | 60 |
| 61 | 62 | 63 | 64 | 65 |
| 66 | 67 | 68 | 69 | 70 |
| 71 | 72 | 73 | 74 | 75 |
| 76 | 77 | 78 | 79 | 80 |
| 81 | 82 | 83 | 84 | 85 |
| 86 | 87 | 88 | 89 | 90 |
| End |
Credits: Based on the excellent class notes provided by, Dr. Lohmeier-Vogel during Fall 2014.
FAQ | Report an Error
You may download this exam as a PDF file here
If you get a question wrong, you can still click on the other answers. This will open up hints and explanations (if available), which will provide additional information.