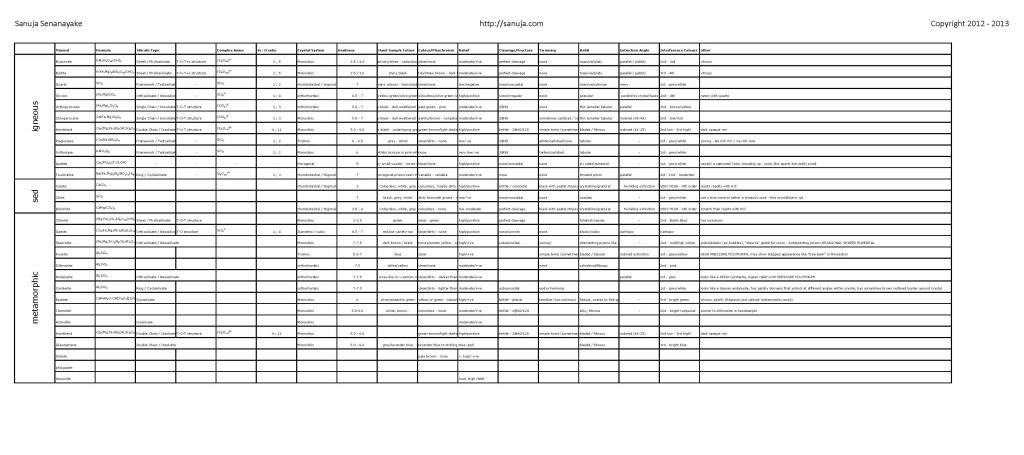
Go to: Midterm Exam
Geology (GLGY 423-UCAL) Final
Some questions have hints and explanations. Click Start to begin.
Start
Congratulations - you have completed Geology (GLGY 423-UCAL) Final.
You scored %%SCORE%% out of %%TOTAL%%.
Your performance has been rated as %%RATING%%
Your answers are highlighted below.
Question 1 |
Polymorphism is caused by differences in thermodynamic properties at different P-T conditions.
A | True |
B | False |
Question 2 |
Alkali feldspars may have several different types of exsolutions. Which of the following type of exsolution can only be observed under X-ray techniques?
A | Microperthite |
B | Macroperthite |
C | Cryptoperthite |
D | Perthite |
E | Antiperthite |
Question 3 |
High temperature and/or dry conditions favors the growth of __I__ crystals over __II__ crystals.
A | I. clinopyroxenes II. orthopyroxenes |
B | I. olivine II. pyroxenes |
C | I. orthopyroxenes II. clinopyroxenes |
D | I. hydrous II. anhydrous |
E | I. pyroxenes II. olivine |
Question 4 |
Which of the following analytical methods should you use for determining the crystal structure of a mineral? (choose the best answer)
A | ICP and AAS |
B | X-ray Diffraction |
C | Electron Microprobe |
D | SEM (Scanning Electron Microscope) |
E | XRF (X-Ray Fluorescence) |
Question 4 Explanation:
While none of the above techniques world provide good details on crystal structures, the X-ray Diffraction is the best choice.
Question 5 |
Which of the following is the most dense mineral group in terms of it's chemical structure?
A | Amphiboles |
B | Pyroxenes |
C | Olivines |
Question 6 |
What is the universally accepted standard thickness of a petrographic thin section?
A | 0.003 mm |
B | 30 000 nm |
C | 0.03 nm |
D | 0.35 mm |
E | 0.30 mm |
Question 6 Explanation:
Remember, 30 000 nm = 0.03 mm
Question 7 |
What is the crystal system and the optic sign of the following crystal?

Image credit: Zoltai and Stout (1985) Mineralogy: Problems and solutions
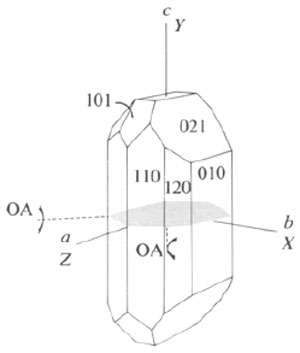
Image credit: Zoltai and Stout (1985) Mineralogy: Problems and solutions
A | Triclinic / negative |
B | Orthorhombic / negative |
C | Monoclinic / positive |
D | Orthorhombic / positive |
E | Monoclinic / negative |
F | Triclinic / positive |
Question 8 |
Given the following information, calculate the 2θ angle.
λ = 0.3419 m
d210 = 3.92564 Å
n = 1
λ = 0.3419 m
d210 = 3.92564 Å
n = 1
A | 11.48 degrees |
B | 4.992 degrees |
C | 6.495 degrees |
D | 22.96 degrees |
E | 2.495 degrees |
Question 8 Explanation:
Don't forget it is asking for 2θ not theta; = 2.495 degrees.

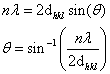
Question 9 |
What are the bottom two end-members of Olivine ternary diagram?
A | Forsterite and Ferrosilite |
B | Forsterite and Faylite |
C | Forsterite and Enstatite |
D | Ferrosilite and Enstatite |
E | Faylite and Enstatite |
F | Ferrosilite and Faylite |
Question 10 |
In X-ray Diffraction Pattern Analysis, the high intensity regions will be recorded as...
A | higher (high amplitude) peaks. |
B | narrower (thinner) peaks. |
C | thicker (fatter) peaks. |
D | lower (low amplitude) peaks. |
Question 11 |
The chemical formula for the Pyroxene iron end-member Orthoferrosilite is Fe2SiO3.
A | False |
B | True |
Question 11 Explanation:
FeSiO3 would be correct.
Question 12 |
Garnets are isotropic minerals.
A | True |
B | False |
Question 12 Explanation:
Not always buddy!
Question 13 |
The following interference diagram can be best described as...

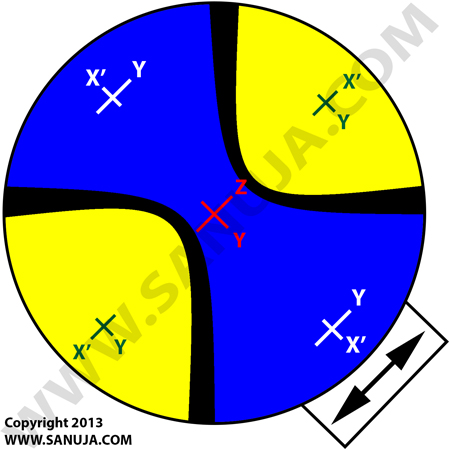
A | an uniaxial Bxa negative. |
B | a biaxial optic axis. |
C | an uniaxial optic axis. |
D | a biaxial Bxa negative. |
E | a biaxial Bxa positive. |
Question 14 |
Minerals with same/similar structure but different compositions is known as...
A | polymorphs |
B | isomorphs |
C | ordered |
D | disordered |
Question 15 |
The "phase-diagrams" are used to describe what type of chemical reactions?
A | Reversible chemical reactions. |
B | Chemical reactions occurred in solid solutions. |
C | Rock forming chemical reactions. Hint: Not the best choice out of the given choices here. |
D | Chemical reactions involving solid materials. |
E | Organic chemical reactions. |
Question 16 |
What is the crystal system and the optic sign of the following crystal?

Image credit: Zoltai and Stout (1985) Mineralogy: Problems and solutions
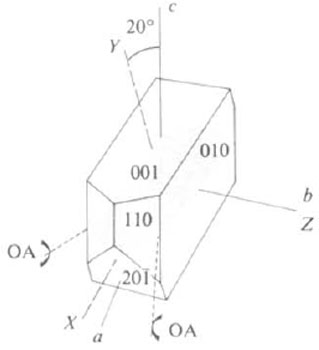
Image credit: Zoltai and Stout (1985) Mineralogy: Problems and solutions
A | Monoclinic / negative |
B | Orthorhombic / negative |
C | Triclinic / positive |
D | Triclinic / negative |
E | Orthorhombic / positive |
F | Monoclinic / positive |
Question 17 |
Which of the following is the best way to identify a mineral?
A | Interference Figure |
B | Chemical composition analysis. |
C | Properties in hand sample (physical properties) |
D | Refractive Index |
E | Birefringence |
Question 18 |
The length fast direction is at ___ degrees to the length slow direction.
A | between 45 - 90 |
B | 45 |
C | between 90 - 180 |
D | 90 |
E | 180 |
Question 19 |
What is the Miller index for 10a: 3b: 5c?
A | (5 3 10) |
B | (3 10 6) |
C | (1/10 1/3 1/5) |
D | (1 5 3) |
E | (10 3 5) |
Question 20 |
Which cut will provide a Bxo figure for the following crystal?

Image credit: Zoltai and Stout (1985) Mineralogy: Problems and solutions
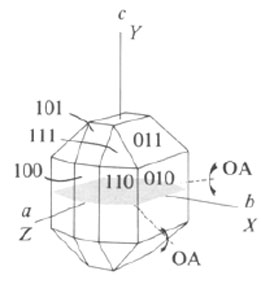
Image credit: Zoltai and Stout (1985) Mineralogy: Problems and solutions
A | 100 |
B | 101 |
C | 111 |
D | 110 |
E | 001 |
Question 21 |
Does (123) plain lines in the [210] zone?
A | No |
B | Yes |
Question 21 Explanation:
No because (1X2) + (2X1) + (3X0) is not equal to zero.
Question 22 |
In plagioclase solid solution composition diagram, the Albite melting occur at ____ Anorthite melting point.
A | high temperatures compared to |
B | low temperatures compared to |
C | same temperature as |
Question 23 |
The analytical methods, ICP and AAS will destroy the tested sample and often the same sample cannot be used for further analysis.
A | False |
B | True |
Question 24 |
The best property to identify feldspars is the unique twining.
A | False |
B | True |
Question 24 Explanation:
"In metamorphic rocks, and in low temperature diagenetic rocks in which secondary feldspars may grow, feldspars are typically untwinned." - Dr. Sytle Antao
Question 25 |
What is the best description for the following effect in Calcite crystals?
A | This is a result of internal diffraction of natural light. |
B | This is an optical property caused by non-visible UV light interacting with the visible light spectrum. |
C | This is an optical property that would only occur in Calcite crystals. Hence it is a good property for mineral identification. |
D | This is caused by the difference in Birefringence. |
E | This is caused by the difference in velocities between the epsilon and omega rays. |
Question 26 |
The chemical formula for the Pyroxene end-member Enstatite is MgSiO3.
A | False |
B | True |
Question 27 |
Electromagnetic radiation is always....
A | constructive |
B | constructive and destructive |
C | confined and non-scattering |
D | destructive |
Question 28 |
The T-O-T offsets structures in clinopyroxenes are situated in such...
A | alternating between positive and negative faces. |
B | all facing the same direction. |
C | longer a - axis compared to the a-axis length of the orthopyroxenes. |
D | randomly arranged T-O-Ts alternates every two chains. |
Question 29 |
Given the following information, calculate the wavelength in nm.
Birefringence: 0.024
Thickness of the slide: 0.03 mm
Birefringence: 0.024
Thickness of the slide: 0.03 mm
A | 2.14 -3 nm |
B | 650 nm |
C | 7.20 x 10-4 nm |
D | 720 nm |
E | 5.60 10-4 nm |
F | 560 nm |
Question 29 Explanation:
Question 30 |
__I__ temperature reactions will result in __II__.
A | I. low II. high symmetry. |
B | I. low II. disordered minerals. |
C | I. high II. ordered minerals. |
D | I. high II. low symmetry. |
E | I. high II. high symmetry. |
Question 31 |
Which of the following planes lines on the [1 1 1] zone?
A | (132) |
B | (122) |
C | (112) |
D | (212) |
E | (121) |
Question 32 |
__I__ are low-T, slow cooled alkali feldspars and their Optic Axis Plane (OAP) is __II__ to 010.
A | I. Microcline and Sanidine II. parallel |
B | I. Microcline and Sanidine II. perpendicular |
C | I. Microcline and Orthoclase II. perpendicular |
D | I. Microcline and Orthoclase II. parallel |
E | I. Sanidine and Orthoclase II. perpendicular |
Question 33 |
Which of the following is the most dense mineral in terms of it's chemical structure?
A | Andalusite |
B | Epidote |
C | Carbonates |
D | Kyanite |
E | Sillimanite |
Question 34 |
Abundance of water in a olivine forming environment will result in formation of serpentine and talc.
A | True |
B | False |
Question 35 |
What is the chemical formula for Anthophyllite?
A | Fe7Si8O22(OH)2 |
B | Ca2Fe5Si8O22(OH)2 |
C | Ca2Mg5Si8O22(OH)2 |
D | Mg7Si8O22(OH)2 |
Question 36 |
Uniaxial minerals can have inclined extinction.
A | True |
B | False |
Question 36 Explanation:
straight and symmetrical only
Once you are finished, click the button below. Any items you have not completed will be marked incorrect.
Get Results
There are 36 questions to complete.
← |
List |
→ |
Return
Shaded items are complete.
| 1 | 2 | 3 | 4 | 5 |
| 6 | 7 | 8 | 9 | 10 |
| 11 | 12 | 13 | 14 | 15 |
| 16 | 17 | 18 | 19 | 20 |
| 21 | 22 | 23 | 24 | 25 |
| 26 | 27 | 28 | 29 | 30 |
| 31 | 32 | 33 | 34 | 35 |
| 36 | End |
Return
You have completed
questions
question
Your score is
Correct
Wrong
Partial-Credit
You have not finished your quiz. If you leave this page, your progress will be lost.
Correct Answer
You Selected
Not Attempted
Final Score on Quiz
Attempted Questions Correct
Attempted Questions Wrong
Questions Not Attempted
Total Questions on Quiz
Question Details
Results
Date
Score
Hint
Time allowed
minutes
seconds
Time used
Answer Choice(s) Selected
Question Text
All done
Need more practice!
Keep trying!
Not bad!
Good work!
Perfect!
You may download this exam as a PDF file here.
Credits: Based on the excellent class notes provided by, Dr. Sytle Antao during Fall 2013.
FAQ | Report an Error.
Point group animation test run
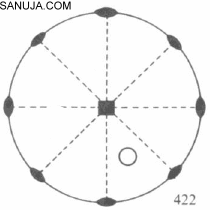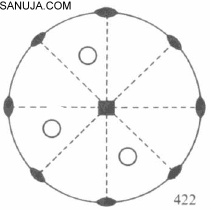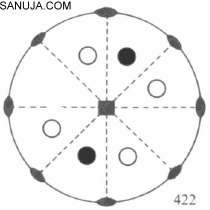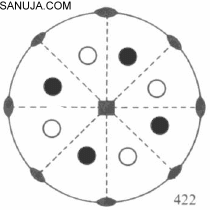
Lab final cheat sheet